Digital Asset Management for Medical Technology
Technical Dokumentation for Medical Devices
We help medical device manufacturers and distributors
manage complex product portfolios consistently, in compliance with legal requirements, and through automated processes across all channels.
- MDR-compliant
- PIM/ERP-Integration
- Dealer portal
- Shorter time to market
- Brand consistency
- Image conversion
Tessa drives top brands in medical technology
Here’s how TESSA supports you and your team in the medical technology sector

Challenge
Technical documentation for medical devices, such as IFUs (Instructions for Use) or data sheets, is constantly updated and stored in various systems. Manually assigning and distributing these files to online stores or distributors is prone to errors and results in outdated versions remaining in circulation.
Your Solution with TESSA
Seamlessly connected systems
By directly connecting TESSA DAM to your PIM system, information from the data sheets and IFUs is imported and synchronized with the product images. This ensures that the most up-to-date, MDR-compliant file version is always available.

Challenge
Product images and related data (eIFU, data sheets, etc.) need to be shared with specialty retailers, distributors, or international locations.
Your Solution with TESSA
Self-Service for Medtech Sales
With TESSA BrandHub, you provide medical technology resellers with controlled access to approved marketing assets. Your partners can download images and the associated information directly in the appropriate target format themselves, without requiring manual intervention on your part.
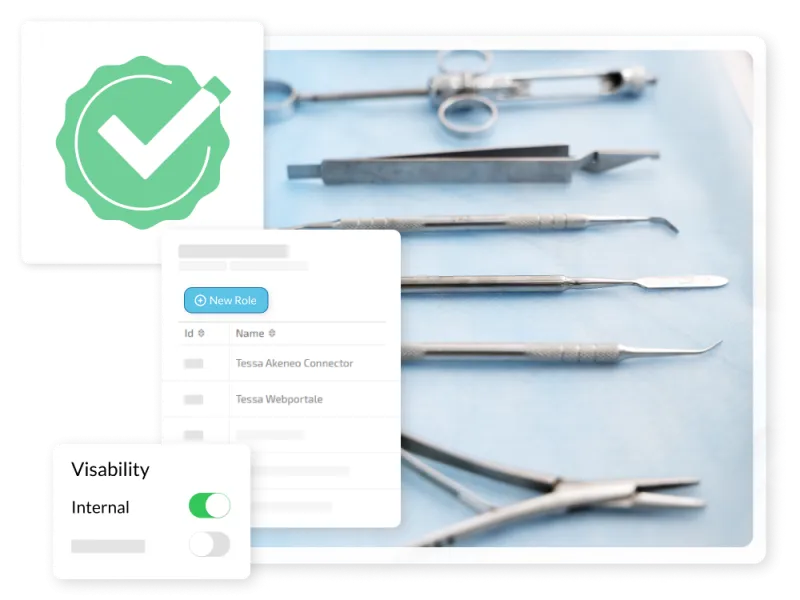
Challenge
Strict MDR requirements mandate that marketing teams and partners access only currently approved materials. Outdated documents in circulation or missing MDR certificates can quickly lead to liability risks for medical device manufacturers.
Your Solution with TESSA
Role & Rights Management
Our DAM precisely controls who sees and uses which content. This ensures a controlled, legally compliant provision of all your assets and the associated UDI labeling for audits.
TESSA inspires users
Additional features of TESSA for medical technology
AI-Feautures
Intelligent AI automation (medical tags, cropping, object recognition, etc.) with TESSA DAM.
Intelligent search
Find the digital assets you need for medical devices easily, intuitively, and quickly.
MedTech-Workflows
Complete tasks in the graphics team faster with our powerful workflow module.